New Study Calls for Re-evaluation of Antidepressant Warnings

In a new study in the American Psychiatric Association’s Psychiatric Research & Clinical Practice, lead and senior authors Christine Lu, PhD, MSc, and Stephen Soumerai, ScD and team probe further into FDA public health advisories issued beginning in late 2003. The advisories cautioned that children, adolescents, and young adults taking antidepressants were at an increased risk of suicidal ideation and behavior. The study team built on their prior work on the same warnings to produce the first long-term study on this subject. We spoke with Drs. Lu and Soumerai to learn more about the study, "Increases in Suicide deaths among Adolescents and Young Adults Following US FDA Antidepressant Boxed Warnings and Declines in Depression Care".
Q: In late 2003, 2005, and 2007, the Food and Drug Administration issued a public health advisory and two severe “black box” warnings that children, adolescents, and young adults taking antidepressants were at an increased risk of suicidal ideation and behavior. These warnings persist today. Can you tell us how the FDA arrived at this conclusion, and what the warnings were intended to do?
A: The FDA issues warnings based on a variety of evidence about drug safety issues, from single case reports to randomized clinical trials.
In this case, there were anecdotal reports of Selective Serotonin Reuptake Inhibitors (SSRIs), or antidepressants, associated with suicidal thoughts and behavior. But here, the direction of causation is unclear: youth taking antidepressants were almost certainly depressed in the first place, sometimes with suicidal thoughts.
Meta-analyses of industry drug trials then found a 1-2% increase in the number of young people experiencing suicidal thinking at the start of their antidepressant therapy. But these meta-analyses are biased: the majority of trials did not have pre-planned suicidality outcomes, and the treatment groups had a significant drop-out rate. There was also no baseline data on SSRI use, which would have observed a reduction in suicide deaths as antidepressant use rose before the warnings, possibly leading to a very different policy.
The goal of the warnings was to alert clinicians to be on the lookout for increased suicidal ideation early in treatment. Unfortunately, the warnings stigmatized mental health care for young people. As care declined, correspondingly, suicide attempts and suicide deaths increased.
Q: Tell us about your journey to study the results of these warnings – what inspired the research, and why have you stuck with it to this day?
A: We realized we had data in the Health Care Systems Research Network (HCSRN) to rigorously address some important unanswered questions about the impacts of the antidepressant warnings. Only a few years after the warnings, several outside scientists began reporting that the antidepressant warnings were causing unintended, large reductions in depression case finding and diagnosis, mental health visits, and antidepressant use among youth, even among those with major depression for whom these represent first-line treatments. And these effects “spilled over” to adults and elderly as well. At least three studies showed that the prior downward trend in youth suicide deaths may have stopped and started increasing. But the later studies only included one or two years (observations) after the initial warnings and were thus only suggestive.
We began work on a large NIMH R01 project in the Mental Health Research Network to study the longer-term consequences of the warnings on changes in trends of antidepressant use and psychotropic drug poisonings (proxy for suicide attempts). Two of these findings published in The BMJ in 2014 are reproduced below. The sudden interruptions in the long baseline trends show that the warnings likely caused underuse of essential care, substantial reductions in antidepressant use, and increases in suicide attempts.
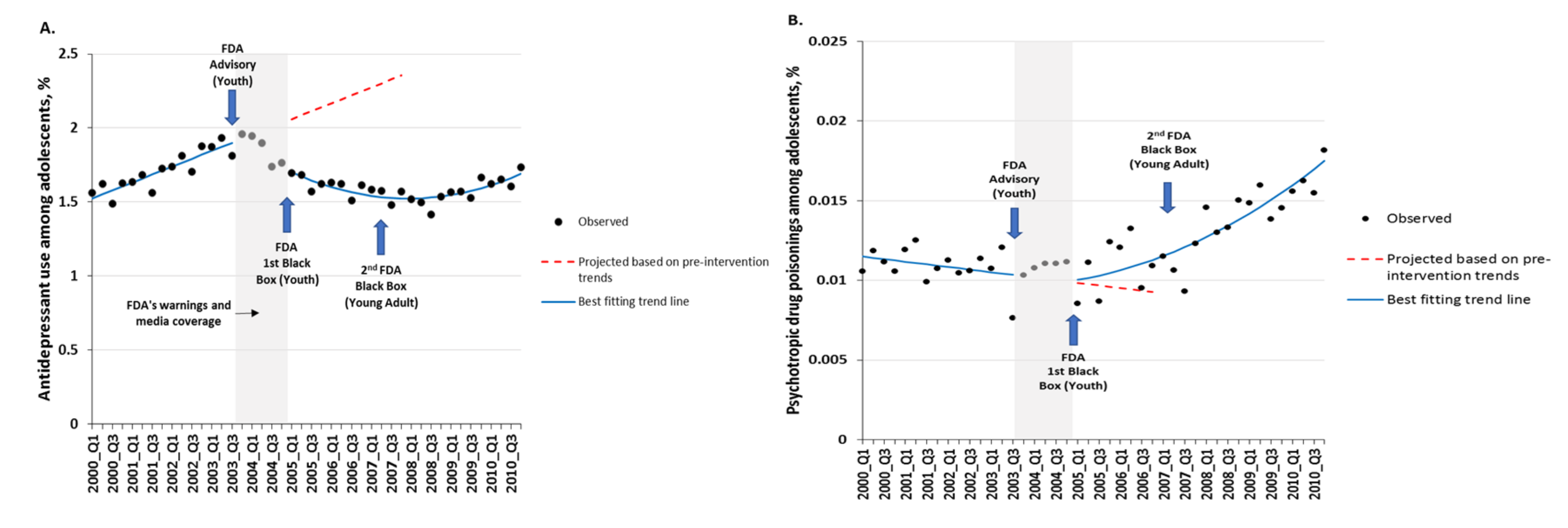
But even the millions of young people in the network were insufficient to study rare suicide deaths (e.g., as low as 4-7 deaths per 100,000 per year). For this study, we collected 28 years of validated CDC U.S. suicide deaths by age to investigate the possible effect of the warnings on suicide deaths among adolescents and young adults. This hypothesis was based on the many previous findings of the warnings’ unintended reductions in mental health care.
Q: Tell us about your findings – did they surprise you?
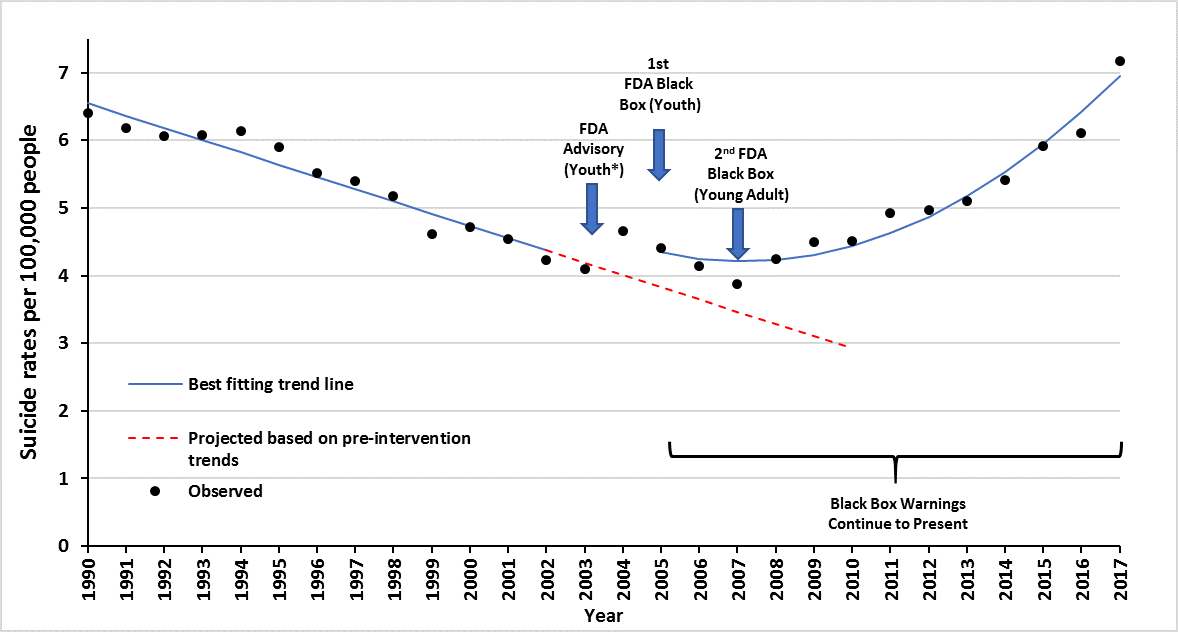
FDA antidepressant warnings.
*Adolescents defined as 10-19 years of age (WHO definition).
Data obtained from the CDC Wonder Database Compressed
Mortality File and Detailed Mortality File.
A: No. We observed large increases in suicide deaths among adolescents and young adults following the widely publicized FDA antidepressant warnings and reductions in mental health care. The figure at left, from the paper, demonstrates the effect (an interruption in the baseline trend line) among adolescents.
The FDA antidepressant advisories, accompanied by media coverage, stigmatized depression care for young people and fostered reluctance by providers to initiate antidepressant treatment. Thus, these warnings reduced diagnosis of major depression, and drug and non-drug mental health treatment.
Other unintentional injury deaths, including opioid deaths, a comparison outcome, did not increase after the warnings. In addition, the “bend in the curve” of suicide deaths did not occur among young middle-aged controls.
Furthermore, 60% to 70% of adolescents and young adults with major depression do not receive any mental health care. While inappropriate use is certainly a concern, the major problem appears to be underuse, not overuse, of all mental health treatments leaving the increased risk from major depression unanswered. These data support a formal re-evaluation of the risks and benefits of the boxed antidepressant warnings, FDA’s strongest type of communications about a drug or drug class.
We recommend the current boxed warnings be lifted entirely or “demoted” to the long list of routine warnings contained in product labeling. The risks of non-treatment and under-treatment of major depression exceed those of the warnings falsely asserting their role in increased suicide among young people.
Q: What is unique about this study?
A: This is the first long-term study to investigate whether suicide deaths in the U.S. increased among adolescents and young adults following the antidepressant warnings and the accompanying declines in depression diagnosis and treatment already demonstrated in large quasi-experimental studies.
Q: You cite that for this particular issue, a randomized trial is infeasible. Tell us how the study was conducted, and why.
A: We used an interrupted times series design, which can provide powerful evidence of policy effects when randomization is not possible, because it measures effects of interruptions in trends soon after interventions, and we controlled for a 13-year stable downward trend in the pre-warning period.
When a nationwide warning like this receives worldwide media attention, it becomes impossible to conduct randomized controlled trials to study the effects.
Q: Your study addresses the increase in suicide deaths, while other studies using the same methods have shown that the warnings caused outcomes that typically precede suicide. Can comparisons be drawn between these studies, especially in relation to the timing and shape of the effects of these studies?
A: Yes. A number of strong interrupted time-series studies (led by Libby, Busch, Kurian, and Gibbons to name a few) showed that the warnings reduced diagnosis of major depression, and drug and non-drug mental health treatment; these are intermediate outcomes that predict suicide. These data add to the evidence of an association between the antidepressant warnings and increased suicide deaths.
The most striking intermediate outcome was the increase in suicide attempts demonstrated by our study in The BMJ in 2014. It is remarkable how similar the post warning trends in attempts are to the suicide deaths in this new study. The findings, from different data, validate one other.